Possible explanation 1: Gel run time is too short. Illustration showing improper protein band separation on an SDS-PAGE gel. The protein bands may also appear as a single broad band, indicating poor resolution.įigure 6. The gel, in this case, might appear blurry with unclear or overlapping protein bands. Improper separation or resolution of your bands in an SDS-PAGE gel could be attributed to several factors like not running the gel long enough, uneven gel concentration, and improper buffer preparation. If you do not need to load all of the wells with experimental samples, load ladders or any other protein in your lab stock. This leads to edge effect on neighboring lanes. 
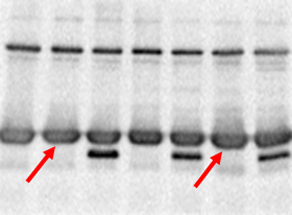
Troubleshooting suggestion: Do not keep wells empty when loading your gel. Possible explanation: Edge effect is due to empty wells. Of the right and left most lanes being distorted. Shows the edge effect of protein bands running on an SDS-PAGE gel as a result This happens when the lanes at the periphery (edge) of the gel are left empty. Often you may find that the right and the left most lanes of your SDS-PAGE gel are distorted due to a phenomenon known as edge effect. Another way is to run the gel at a lower voltage for a longer time. Some options would be to run your gel in a cold room or put ice packs inside the gel-running apparatus. 
You will want to minimize heat production as much as possible. Troubleshooting suggestion: Heat generation is an unwanted side-effect of the electric current flowing through the apparatus. Possible explanation: High heat generated during protein electrophoresis. An illustration of a smiling shape seen in the bands of your This phenomenon is often described as smiling due to the curved appearance of the protein bands.įigure 4. If too much heat is applied during preparation or running your acrylamide gels, the gels may expand, leading to uneven protein band migration. Illustrates the dye front reaching the end of the gel, which is generally a good time to stop running your protein gel. In fact, for resolving very high molecular weight proteins, a high run time may be required.īut in general, the dye front reaching the bottom of the gel is a good time to stop the current.įigure 3. If the molecular weight is high, then you might be fine running the gel even after the dye front has run off the gel. However, this might change depending on the molecular weight of your target protein. Troubleshooting suggestion: A common standard is to stop running the gel when the dye front reaches the bottom of the gel. Possible explanation: You might have run the gel longer than required. The bottom three bands of the ladder (L) along with the lower two bands of samples 1 and 2 have run off the gel pictured on the right. On the right, the lower molecular weight bands ran off the gel. The gel on the left has not been over run. One possible cause is that you ran the gel longer than required.Ī good confirmation of whether or not this has happened, even before staining your gel with coomassie, is by seeing whether the protein ladder – which generally has colored bands, has also run off the gel.įigure 2. If you observe that your protein samples ran off the gel, the gel will appear blank in the region where the protein samples were expected to migrate. On another note, running your gel at a high voltage might also cause the running buffer and your gel to warm up too quickly, which is something to avoid. 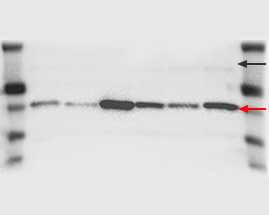
Using a lower voltage for a higher run time gives good results. The bigger your gel, the higher the voltage you need to run it at. Troubleshooting suggestion: A good practice is running the gel at 10-15 Volts/ cm. Possible explanation: You are running your gel at too high of a voltage Illustration of smeared bands on an SDS-PAGE gel. Smeared bands while running an SDS-PAGE gel might occur if the voltage you are running the gel at is too high.įigure 1. Protein samples migrating out of the wells before your SDS-PAGE has started running Rate of protein sample migration in your SDS-PAGE gel is too fast Protein in your sample not properly separated or resolved in your SDS-PAGE gel And for each of these situations, we will provide the troubleshooting steps that might solve the problem.īands in the periphery of your SDS-PAGE gel are distorted We will provide analytical insight into what might have gone wrong. In this article, we will discuss these issues that commonly occur during experimentation while running your protein sample in an SDS-PAGE gel. Samples migrating off the well before running the gel.Rate of samples migrating in your gel is too fast.Bands are not properly separated or resolved.Bands in the periphery of your gel are distorted.Some of these include smeared bands, smiling bands, improper separation, and protein samples migrating off the well before it runs. There are seven different indications of problems with SDS-PAGE gel running. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
